Draw A Picture Of A Set Of P Orbitals
Draw A Picture Of A Set Of P Orbitals - Web draw a picture that shows all three. D orbitals are described only in terms. Web just as with the s orbitals, the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases. The second picture shows the result of the constructive (or destructive) interference. This means that we can. The shapes of the 90% probability. At the same time, we cannot name the new orbitals s or p, for they’re. For a p orbital, draw a figure eight; In the blue box, draw a picture of an s orbital. Web while electron shells and orbitals are closely related, orbitals provide a more accurate picture of the electron configuration of an atom. Web figure 1.4 representations of s, p, and d orbitals. We classified the different orbital into shells and sub shells to distinguish them more easily. Web just as with the s orbitals, the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases. Ypx = (3/4π)1/2 sin θ cos φ = (3/4π)1/2. Of the four, we'll be concerned. Web an orbital is a space where a specific pair of electrons can be found. Web draw a picture that shows all three. What we’ve drawn here is a surface of constant probability. For an f orbital, see below. At the same time, we cannot name the new orbitals s or p, for they’re. The shapes of the 90% probability. Web draw a picture that shows all three. Web just as with the s orbitals, the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases. Web figure 1.4 representations of. The shapes of the 90% probability. Web atomic orbitals are the wavefunctions which are solutions of the schroumldinger equation for the hydrogen atom the subset of atomic orbitals and are plotted in three dimensions. Orbitals on one atom and all three. Ypx = (3/4π)1/2 sin θ cos φ = (3/4π)1/2 x/r. Web 68.draw the structure of molecular orbital of o2. Web the illustration above is for one set of p orbitals that are orthogonal to the bond axis. Ypz = (y0 = (3/4π)1/2 cos θ = (3/4π)1/2 z/r. This is called a p (pi) bond. It explores s and p orbitals in some detail, including their shapes and energies. Draw a picture of a set of p orbitals. Web draw a picture that shows all three 2p orbitals on one atom and all three 2p orbitals on another atom. Web an orbital is a space where a specific pair of electrons can be found. Ypx = (3/4π)1/2 sin θ cos φ = (3/4π)1/2 x/r. Web draw a picture that shows all three. The second picture shows the result. Click here:point_up_2:to get an answer to your question :writing_hand:draw. The second picture shows the result of the constructive (or destructive) interference. Web just as with the s orbitals, the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases. Of the four, we'll be concerned. In the blue box, draw a picture. Make sure you check here first.what quantum numbers can apply to. Web an orbital is a space where a specific pair of electrons can be found. We classified the different orbital into shells and sub shells to distinguish them more easily. Of the four, we'll be concerned. At the same time, we cannot name the new orbitals s or p,. Web atomic orbitals are the wavefunctions which are solutions of the schroumldinger equation for the hydrogen atom the subset of atomic orbitals and are plotted in three dimensions. Web draw a picture that shows all three 2p orbitals on one atom and all three 2p orbitals on another atom. For a p orbital, draw a figure eight; You'll get a. Web draw a picture that shows all three. Web just as with the s orbitals, the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases. Orbitals on one atom and all three. How many antibonding orbitals, and of what type, can be. This is called a p (pi) bond. Web this page discusses atomic orbitals at an introductory level. Web 68.draw the structure of molecular orbital of o2. Make sure you check here first.what quantum numbers can apply to. Web figure 1.4 representations of s, p, and d orbitals. Draw a picture of a set of p orbitals. D orbitals are described only in terms. How many antibonding orbitals, and of what type, can be. Orbitals on one atom and all three. For an f orbital, see below. Click here:point_up_2:to get an answer to your question :writing_hand:draw. Ypx = (3/4π)1/2 sin θ cos φ = (3/4π)1/2 x/r. Web the illustration above is for one set of p orbitals that are orthogonal to the bond axis. Web just as with the s orbitals, the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases. Web below is three plots showing what the \(2p\) orbitals look like. Of the four, we'll be concerned. At the same time, we cannot name the new orbitals s or p, for they’re.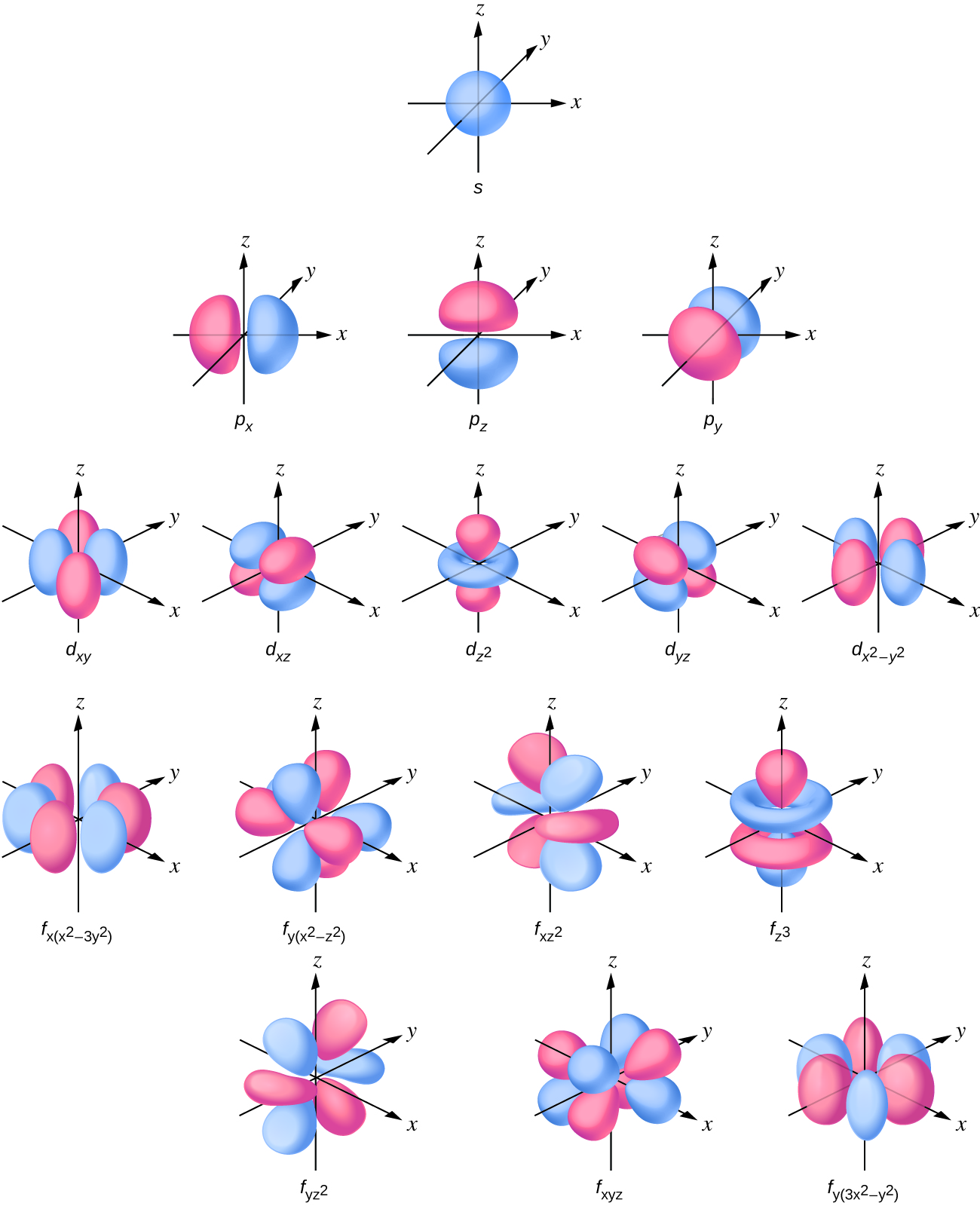
8.3 Development of Quantum Theory CHEM 1114 Introduction to Chemistry
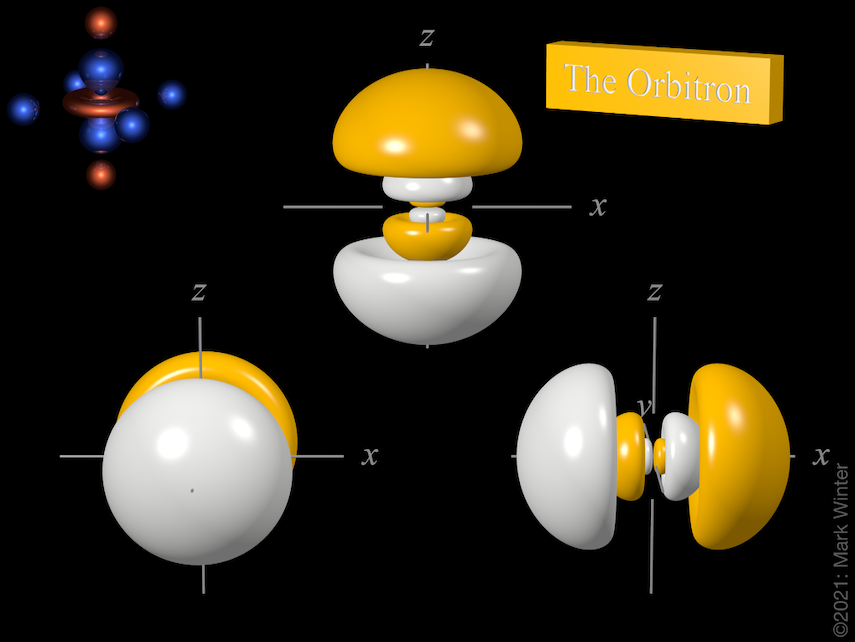
The Orbitron 4p atomic orbitals

How To Draw Orbitals Deepcontrol3
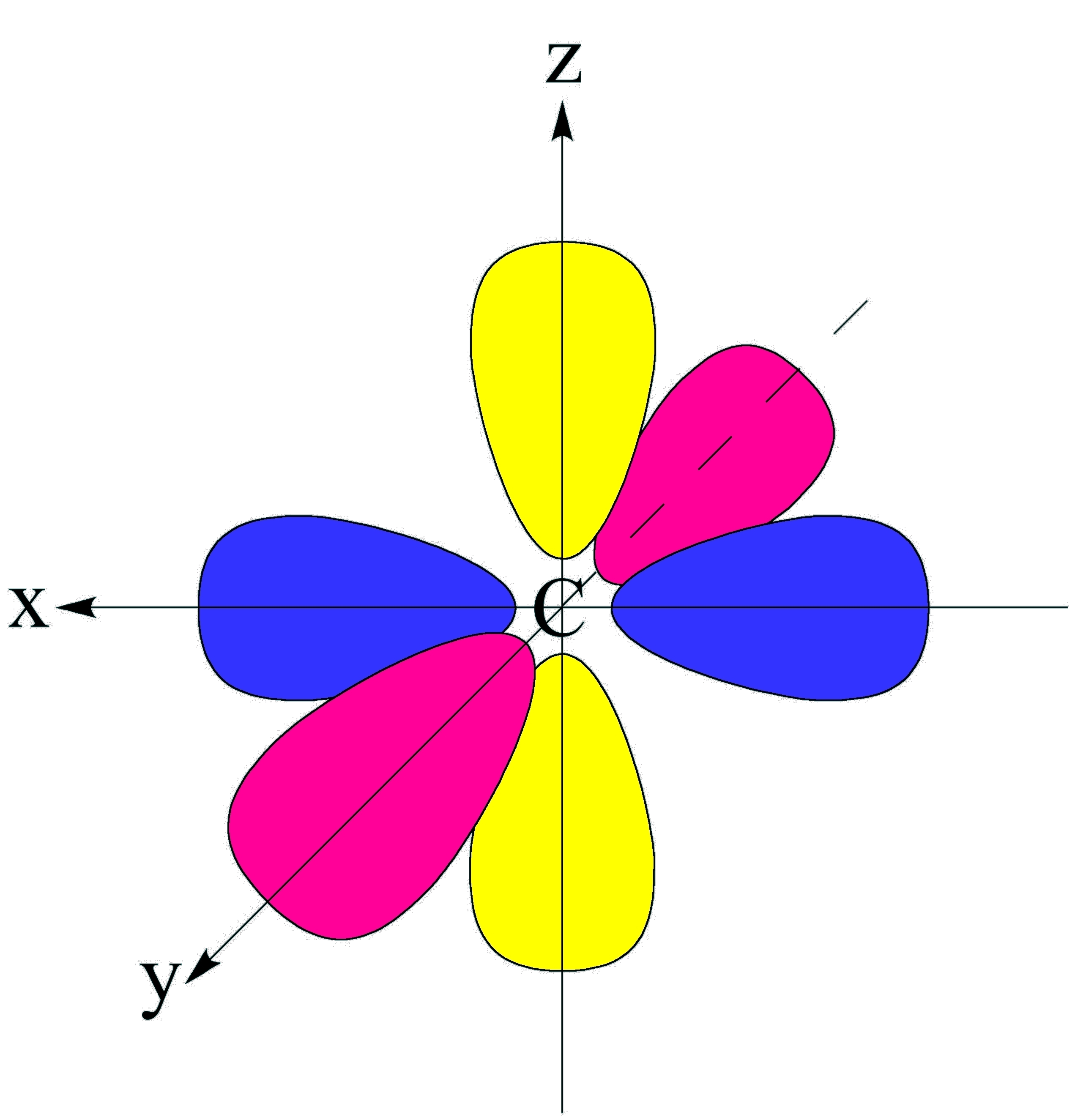
Illustrated Glossary of Organic Chemistry Orbital

Biochemistry Glossary s & p Orbitals Draw It to Know It
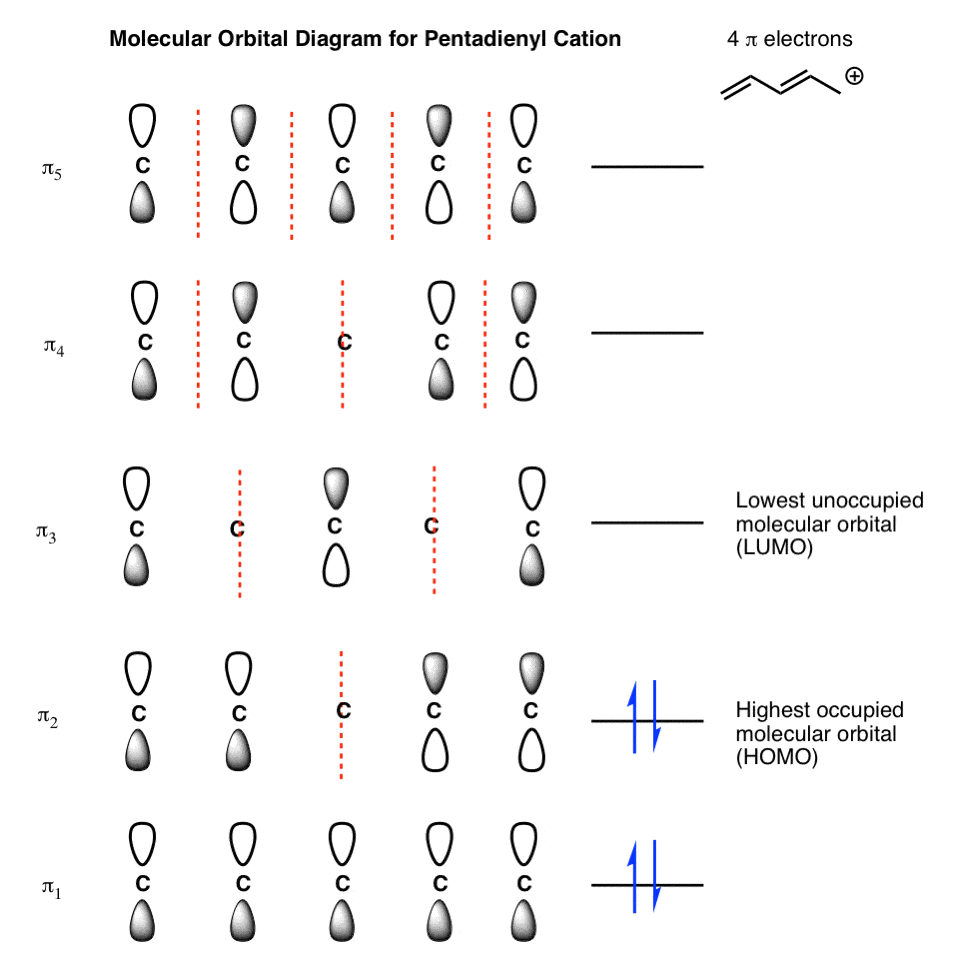
The Pi Molecular Orbitals of Butadiene And How To Draw Them

How do you draw s,p,d,f orbitals? Socratic
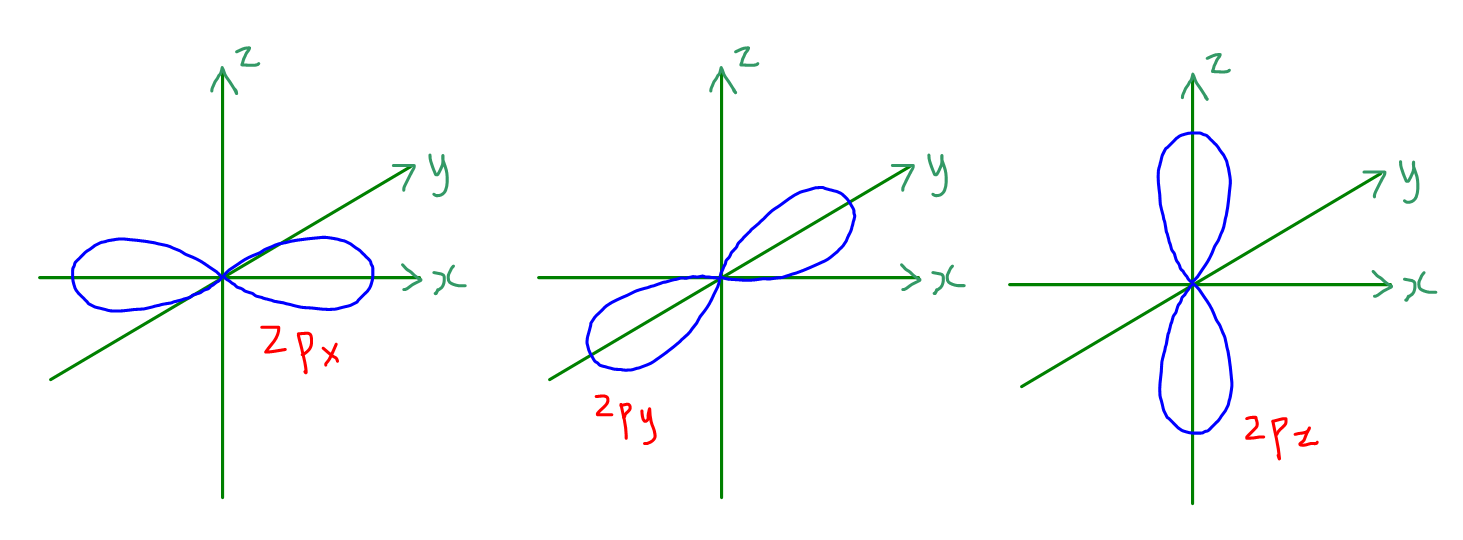
How to Draw Shapes of Orbitals

Molecular Orbitals Introductory Chemistry
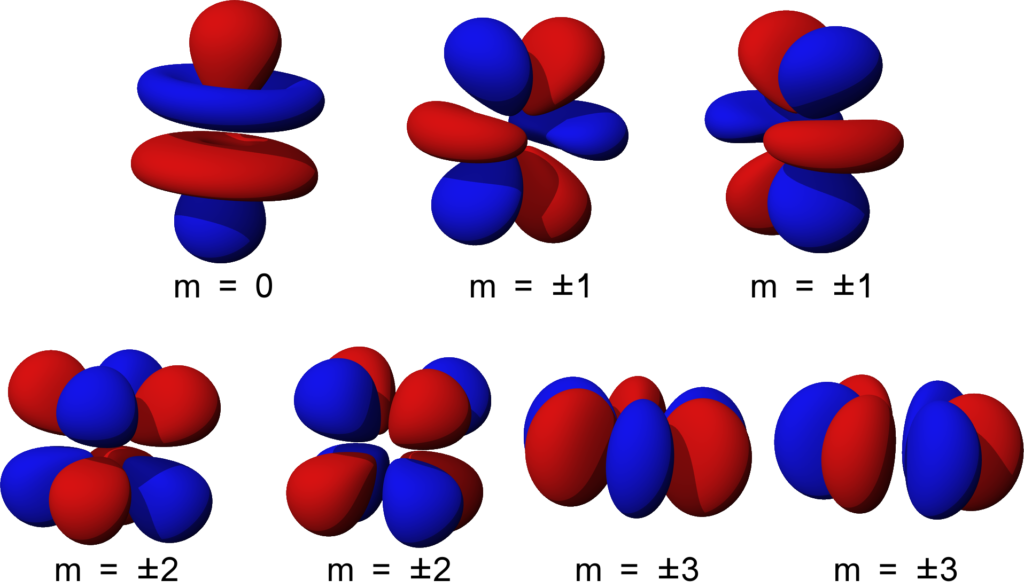
Shapes of Orbitals and their Types Chemistry Skills
For A P Orbital, Draw A Figure Eight;
Web Just As With The S Orbitals, The Size And Complexity Of The P Orbitals For Any Atom Increase As The Principal Quantum Number N Increases.
We Classified The Different Orbital Into Shells And Sub Shells To Distinguish Them More Easily.
Web Draw A Picture That Shows All Three 2P Orbitals On One Atom And All Three 2P Orbitals On Another Atom.
Related Post: